Overview
Professor's question distribution: ≈3 from Ch 27, 3 from Ch 28, 3 from Ch 29, 1-2 from Ch 30, plus 1-2 from the Diffraction lab. About 11-13 questions total.
Module map
- Ch 27 Quantum: blackbody (Wien), photoelectric, X-ray production & Bragg, Compton scattering, de Broglie waves, Heisenberg uncertainty
- Ch 28 Atomic: Bohr energies/radii, hydrogen spectrum (Lyman/Balmer/Paschen), four quantum numbers + Pauli, characteristic X-rays with $Z_\text{eff}$
- Ch 29 Nuclear: nucleus size, activity, exponential decay, half-life, Q values, threshold energy
- Ch 30 Nuclear Energy: fission, fusion, mass-energy conservation
- Lab Diffraction: single-slit $y_m=m\lambda L/a$ and double-slit $y_m=(m+\tfrac12)\lambda L/d$, plus the classic "find $\lambda$ from measured fringe spacing" calc
Study strategy
- Memorize the constants & the $hc=1240$ eV·nm shortcut. Half of Ch 27 collapses into one-line problems with it.
- Know Bohr cold: $E_n = -13.6/n^2$ eV and $r_n = 0.0529 n^2$ nm. Every Ch 28 problem starts here.
- Decay law: $N = N_0(\tfrac12)^{t/T_{1/2}}$ is faster than the $e^{-\lambda t}$ form for textbook half-life problems.
- Compton: $\Delta\lambda$ depends ONLY on $\theta$, not on $\lambda_0$. This trips people up.
- Drill the 10 problems on the Problems tab — they mirror the test format.
- Cheat sheet: build it from the printable template (Cheat Sheet tab) the night before.
Constants & shortcuts (always on hand)
| Symbol | Value | Use it for |
|---|---|---|
| $h$ | $6.626\times10^{-34}$ J·s = $4.136\times10^{-15}$ eV·s | photon energy, de Broglie |
| $\hbar = h/2\pi$ | $1.055\times10^{-34}$ J·s | uncertainty, angular momentum |
| $c$ | $3.00\times10^{8}$ m/s | everything light-related |
| $e$ | $1.602\times10^{-19}$ C | 1 eV = $1.602\times10^{-19}$ J |
| $m_e$ | $9.11\times10^{-31}$ kg | de Broglie, Compton, Bohr |
| $m_e c^2$ | $0.511$ MeV | Compton, relativistic KE |
| $m_p c^2$ | $939$ MeV | nuclear, threshold |
| $hc$ | $1240$ eV·nm | photon E in eV from $\lambda$ in nm — memorize |
| $\lambda_C = h/m_e c$ | $2.43\times10^{-12}$ m | Compton shift max = $2\lambda_C$ |
| $1$ u (atomic mass unit) | $931.494$ MeV/$c^2$ | fission/fusion energy from mass defect |
| $r_0$ | $1.2\times10^{-15}$ m | nucleus radius $r=r_0 A^{1/3}$ |
| $0.0529$ nm | Bohr radius $a_0$ | $r_n = a_0 n^2$ |
Ch 27 — Quantum Physics
1. Blackbody Radiation & Wien's Law
A blackbody absorbs all radiation and re-emits a thermal spectrum that depends only on temperature.
Wien's displacement law — peak wavelength
- Hotter object → shorter peak wavelength (blue-shift).
- Used to estimate stellar surface temperatures from their color.
The UV catastrophe and Planck's quantization
Classical Rayleigh–Jeans predicted energy density $\to\infty$ as $\lambda\to 0$. Reality: drops off. Planck quantized oscillator energies:
Energy is exchanged in discrete packets of size $hf$. This single fix launched quantum mechanics.
Photon intensity (number per second per area)
Where $n$ = photons per unit area per unit time, $hf$ = energy per photon.
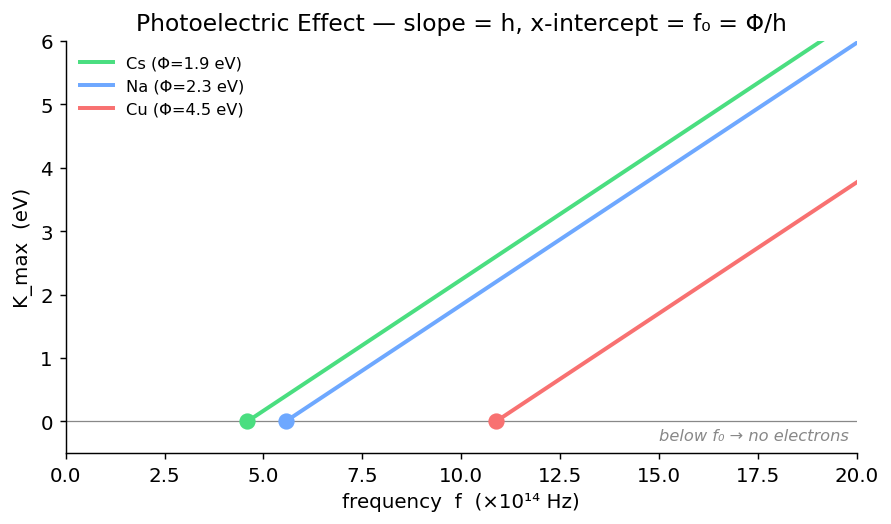
2. Photoelectric Effect
Shine light on a metal → electrons get ejected (photoelectrons). Classical wave theory cannot explain the observations; Einstein's photon model can.
Einstein's photoelectric equation
- $\Phi$ = work function (eV) — minimum energy to free an electron from the metal.
- $V_s$ = stopping potential — reverse voltage that just stops the fastest electrons.
Threshold frequency / wavelength (no emission below)
What classical theory CANNOT explain
- A threshold frequency exists — below $f_0$, no electrons regardless of intensity.
- $K_\text{max}$ depends on frequency, not intensity.
- Intensity changes the number of electrons, not their speed.
- Emission is essentially instantaneous (no energy build-up time).
Common metal work functions
| Metal | $\Phi$ (eV) |
|---|---|
| Cs | 1.9 |
| Na | 2.3 |
| Cu | 4.5 |
| Pt | 5.6 |
3. X-rays — Production & Bragg Diffraction
Two kinds of X-ray spectra
- Bremsstrahlung ("braking radiation"): continuous spectrum from decelerating electrons hitting the anode.
- Characteristic X-rays: sharp lines when an inner-shell vacancy is filled. (See Ch 28 for $Z_\text{eff}$ formula.)
Minimum wavelength (max photon energy)
One electron loses ALL its kinetic energy ($e\Delta V$) in a single collision → highest-energy photon.
Bragg's law — X-ray diffraction from a crystal
Parallel rays reflect off adjacent crystal planes spaced $d$ apart. The extra path of the lower ray is $2d\sin\theta$. Constructive interference requires that = $m\lambda$.
4. Compton Scattering
X-ray scatters off a (nearly) free electron. The scattered photon has longer wavelength — energy went into electron recoil. Only makes sense if the photon is a particle with momentum.
Compton formula
$\lambda_C = h/m_e c = 2.43\times10^{-12}$ m is the Compton wavelength.
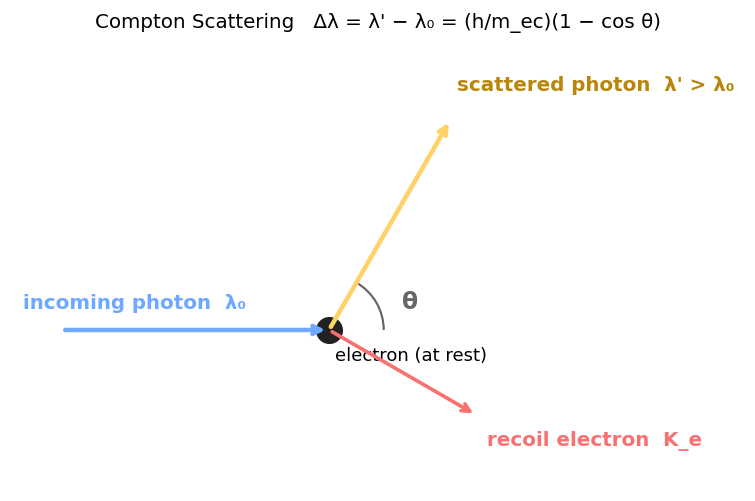
Special angles
| $\theta$ | $1-\cos\theta$ | $\Delta\lambda$ |
|---|---|---|
| $0°$ | 0 | 0 (no shift) |
| $90°$ | 1 | $\lambda_C = 2.43$ pm |
| $180°$ (back-scatter) | 2 | $2\lambda_C = 4.86$ pm (max) |
Electron kinetic energy after the collision
5. de Broglie Matter Waves
If light (waves) can act as particles, then particles act as waves:
Electron accelerated through voltage $V$
From $eV = \tfrac12 m_e v^2 \Rightarrow p = \sqrt{2 m_e eV}$:
$V = 150$ V → $\lambda \approx 0.1$ nm = atomic spacing → electron diffraction (Davisson–Germer experiment, 1927).
6. Heisenberg Uncertainty Principle
These are fundamental, not experimental. A particle does not have a simultaneous exact position and momentum.
Why electrons don't collapse into the nucleus
Confine an electron to atomic size ($\Delta x \sim 10^{-10}$ m): $\Delta v \sim 10^6$ m/s. The kinetic energy needed to confine it more would exceed Coulomb attraction.
Ch 28 — Atomic Physics
1. Bohr Model — Energies and Radii of Hydrogen
Allowed energies
- $n = 1$ → $E_1 = -13.6$ eV (ground state)
- $n = 2$ → $E_2 = -3.40$ eV
- $n = 3$ → $E_3 = -1.51$ eV
- $n \to \infty$ → $E = 0$ (electron just barely free, ionization complete)
Allowed radii
$n = 1$: $r_1 = 0.0529$ nm = the Bohr radius $a_0$. Atom gets bigger as $n^2$.
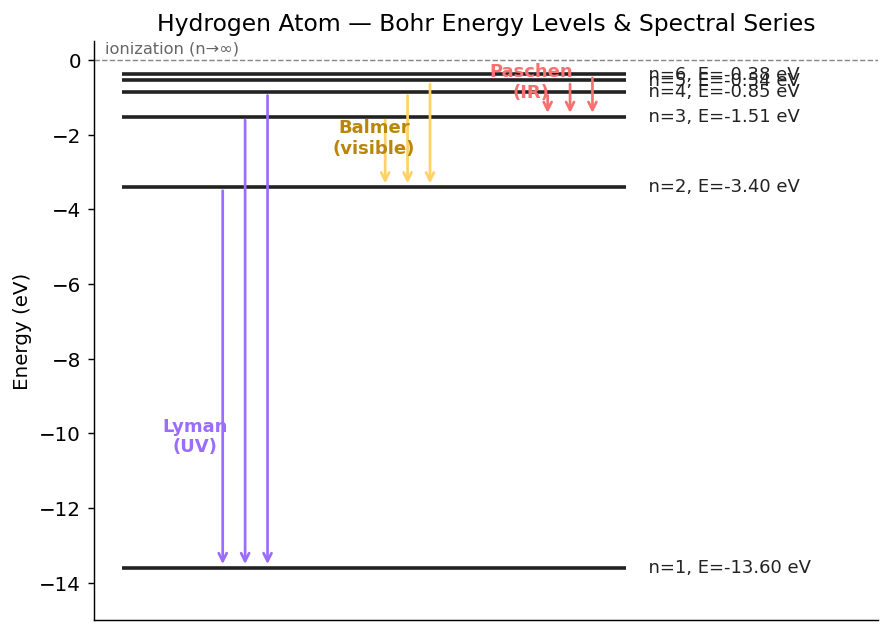
2. Hydrogen Emission Spectrum
When the electron jumps from $n_i$ down to $n_f$, it emits a photon of energy:
(Sign carefully: $1/n_i^2 - 1/n_f^2$ is negative when $n_i > n_f$ — but the photon energy is positive. The formula sheet writes both terms with $-13.6$ in front so the difference comes out positive.)
Named spectral series
| Series | $n_f$ | Region | Notable line |
|---|---|---|---|
| Lyman | 1 | UV | $n=2 \to 1$: $\lambda = 121.6$ nm |
| Balmer | 2 | visible | $n=3 \to 2$ (H$_\alpha$): $\lambda = 656.3$ nm (red) |
| Paschen | 3 | IR | $n=4 \to 3$: $\lambda = 1875$ nm |
3. Four Quantum Numbers & Pauli Exclusion
An electron in an atom is described by 4 quantum numbers:
| Symbol | Name | Allowed values | # of states |
|---|---|---|---|
| $n$ | principal | $1, 2, 3, \dots$ | any positive integer |
| $\ell$ | orbital | $0, 1, 2, \dots, n-1$ | $n$ values |
| $m_\ell$ | orbital magnetic | $-\ell, -\ell+1, \dots, +\ell$ | $2\ell+1$ values |
| $m_s$ | spin magnetic | $+\tfrac12, -\tfrac12$ | 2 values |
Pauli Exclusion Principle
No two electrons in an atom can have the same set of all four quantum numbers. This is what fills the periodic table — each shell holds at most $2n^2$ electrons.
| Shell ($n$) | Subshells ($\ell$) | Max electrons ($2n^2$) |
|---|---|---|
| 1 (K) | s | 2 |
| 2 (L) | s, p | 8 |
| 3 (M) | s, p, d | 18 |
| 4 (N) | s, p, d, f | 32 |
4. Characteristic X-rays (Bohr with $Z_\text{eff}$)
An incoming high-energy electron knocks out an inner-shell electron. An outer-shell electron drops down to fill the vacancy and emits a high-energy photon — this is the characteristic X-ray. The frequency obeys Bohr's formula but with $Z_\text{eff}^2$ replacing the $1$ for hydrogen:
For a $K_\alpha$ X-ray (transition $n=2 \to 1$ with one remaining $K$-shell electron screening): $Z_\text{eff} \approx Z - 1$.
Ch 29 — Nuclear Physics
1. Nucleus Size
Nuclei are approximately spherical with average radius:
$A$ = mass number (total nucleons). Volume $\propto A$ → constant nuclear density.
Notation
$_Z^A X$ means nucleus $X$ with $Z$ protons and $A$ total nucleons (so $N = A - Z$ neutrons).
Example: $^{235}_{92}\text{U}$ — uranium with 92 protons, 235 nucleons (143 neutrons).
2. Activity, Decay Constant, Exponential Decay
Activity (decay rate)
$\lambda$ = decay constant (per second). Units of $R$: becquerels (Bq) = decays/sec.
Number of nuclei vs time
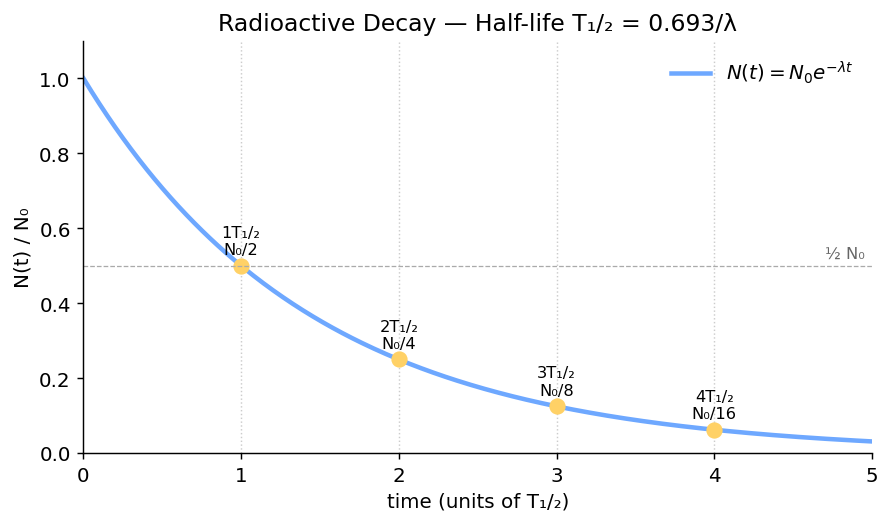
3. Half-life
Time for half the nuclei to decay:
Easy "n half-lives" form
This form is faster than the exponential one when $t$ is a clean multiple (or fraction) of $T_{1/2}$. Example: after 3 half-lives, $N = N_0/8$.
4. Q values, Endothermic vs Exothermic, Threshold Energy
Q value
Energy released in a nuclear reaction:
- Q > 0: exothermic — energy released, can occur spontaneously (energetically).
- Q < 0: endothermic — incoming particle must supply at least enough KE.
Threshold energy (for endothermic reactions)
To conserve both energy AND momentum, the incident particle needs more KE than just $|Q|$:
$m$ = mass of incident particle, $M$ = mass of target. The extra factor accounts for kinetic energy carried off by the recoiling system.
Ch 30 — Nuclear Energy & Elementary Particles
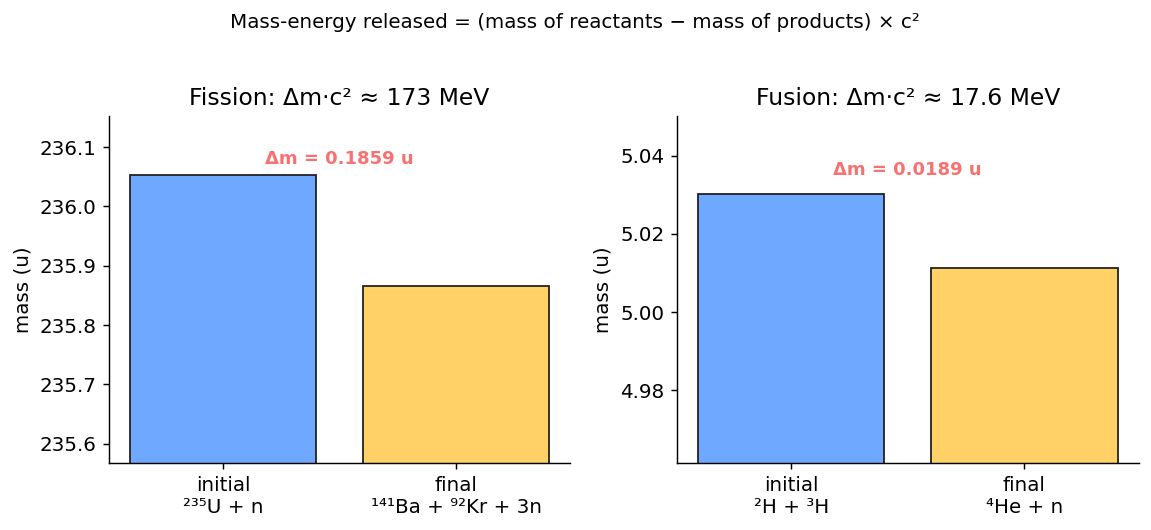
1. Mass-Energy Conservation
Both fission and fusion: the total mass of the products is less than the original mass of the reactants. The missing mass becomes energy:
Where $E_R = mc^2$ is the rest energy of each species, and $\Delta m$ is the total mass defect.
2. Nuclear Fission
A heavy nucleus splits into two smaller nuclei, releasing $\sim 200$ MeV per event.
Classic example: U-235 + neutron
The 3 emitted neutrons can trigger more fissions → chain reaction. This is the basis of nuclear reactors and weapons.
Why fission releases energy
The binding-energy-per-nucleon curve peaks near iron ($A \approx 56$). Splitting a much heavier nucleus moves products toward this peak → more tightly bound → energy released.
3. Nuclear Fusion
Two light nuclei combine to form a heavier nucleus, also releasing energy.
D-T fusion (the easiest reaction; powers stars and tokamaks)
Why fusion is hard
- Both reactants are positively charged → enormous Coulomb barrier.
- Need extreme temperatures ($10^7$ – $10^8$ K) so nuclei have enough KE to tunnel through the barrier.
- The Sun fuses hydrogen into helium via the proton-proton chain at $\sim 1.5\times10^7$ K core temperature.
4. Quick particle-physics conventions
Conservation laws to remember
- Energy (including mass-energy)
- Momentum
- Charge
- Mass number $A$ (nucleons in/out balance)
- Atomic number $Z$ (protons in/out balance)
If a problem asks "is this reaction possible?", check that $A$ and $Z$ are conserved AND that $Q \ge 0$ (or threshold KE supplied).
Lab — Diffraction & Interference
The lab measured the wavelength of a 638 nm laser by examining diffraction patterns from single and double slits at slit-to-screen distance $L \approx 3.5$ m. Expect 1-2 exam questions covering the formulas below and the wavelength-determination calculation.
1. Single-Slit Diffraction
Position of the $m^\text{th}$ dark fringe
$a$ = slit width, $L$ = slit-to-screen distance.
Width of bright fringes
- Central bright $w_0 = 2\lambda L/a$ (twice as wide as the others — spans $m=-1$ to $m=+1$)
- Higher-order brights $w_1 = w_2 = \lambda L / a$
- Ratio $w_0 / w_1 = 2$, $w_1/w_2 = 1$
Doubling the slit width
$a$ in the denominator → all widths get cut in half. Wider slit = narrower envelope.
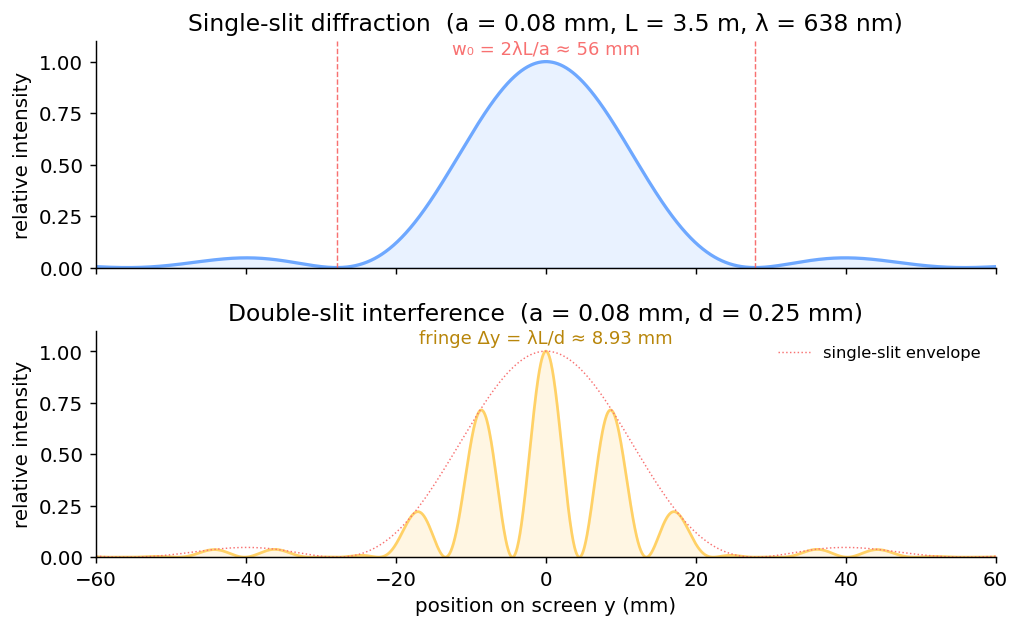
2. Double-Slit Interference
Position of the $m^\text{th}$ dark fringe
$d$ = slit spacing (center-to-center).
Bright fringe spacing
All bright fringes (including the central) have the same width $\lambda L / d$. Pattern is uniformly spaced — unlike single slit.
Distance between $\pm 5^\text{th}$ dark fringes (lab Q)
Counting outward from center: 1st dark at $\tfrac12 \lambda L/d$, 5th dark at $\tfrac{9}{2}\lambda L/d$. Distance between $+5^\text{th}$ and $-5^\text{th}$:
Doubling the slit spacing
$d$ in the denominator → fringe spacing halves. Pattern becomes denser (more fringes packed in).
3. Determining $\lambda$ from measured fringe spacing
From single-slit central-bright width
Solve $w_0 = 2\lambda L/a$ for $\lambda$:
From double-slit fringe spacing
Solve $D_5 = 9\lambda L/d$ for $\lambda$:
Worked example (from the lab)
Single slit: $a = 0.04$ mm, $L = 3.50$ m, measured $w_0 = 110$ mm:
$\lambda = (0.110)(0.04\times10^{-3}) / (2 \times 3.50) = 6.29\times10^{-7}$ m = 629 nm
Compare to actual 638 nm → 1.4% error. ✓
Formula Sheet — Print-Ready
Use Cmd/Ctrl+P. Only this tab prints. Designed to fit on 1-2 letter pages.
Constants
| Symbol | Value |
|---|---|
| $h$ | $6.63\times10^{-34}$ J·s = $4.14\times10^{-15}$ eV·s |
| $c$ | $3.00\times10^{8}$ m/s |
| $hc$ | 1240 eV·nm |
| $e$ | $1.60\times10^{-19}$ C |
| $m_e$ | $9.11\times10^{-31}$ kg, $m_e c^2 = 0.511$ MeV |
| $m_p$ | $1.67\times10^{-27}$ kg, $m_p c^2 = 939$ MeV |
| $\lambda_C$ | $h/m_e c = 2.43$ pm |
| $1$ u | $931.494$ MeV/$c^2$ |
| $r_0$ (nucleus) | $1.2\times10^{-15}$ m |
| $a_0$ (Bohr) | $0.0529$ nm |
Ch 27 Quantum
| Quantity | Formula | Notes |
|---|---|---|
| Wien's law | $\lambda_\text{max} T = 2.898\times10^{-3}$ m·K | peak of blackbody curve |
| Photon intensity | $I = n\, hf$ | $n$ = photons / area / sec |
| Photon energy | $E = hf = hc/\lambda$ | $E(\text{eV}) = 1240/\lambda(\text{nm})$ |
| Photoelectric | $K_\text{max} = hf - \Phi = eV_s$ | Einstein |
| Threshold | $f_0 = \Phi/h,\ \lambda_0 = hc/\Phi$ | no emission below |
| X-ray min $\lambda$ | $\lambda_\text{min} = hc/(e\Delta V)$ | $\Delta V$ = tube voltage |
| Bragg's law | $2d\sin\theta = m\lambda$ | $\theta$ from plane |
| Compton shift | $\Delta\lambda = (h/m_e c)(1-\cos\theta)$ | independent of $\lambda_0$ |
| de Broglie | $\lambda = h/p = h/(mv)$ | all matter |
| de Broglie e⁻ | $\lambda(\text{nm}) \approx 1.226/\sqrt{V}$ | $V$ in volts |
| Uncertainty | $\Delta x \cdot \Delta p \ge h/4\pi$ | $\Delta E \cdot \Delta t \ge h/4\pi$ |
Ch 28 Atomic
| Quantity | Formula | Notes |
|---|---|---|
| Energy levels (H) | $E_n = -13.6/n^2$ eV | $n=1,2,3,\dots$ |
| Bohr radii (H) | $r_n = (0.0529\text{ nm})\, n^2$ | $r_1 = a_0$ |
| Emission spectrum | $hf = (-13.6\text{ eV})(1/n_i^2 - 1/n_f^2)$ | $\lambda = hc/(\Delta E)$ |
| Quantum numbers | $n,\ell, m_\ell, m_s$ | Pauli: no two same set |
| Allowed $\ell$ | $0, 1, \dots, n-1$ | $2\ell+1$ values of $m_\ell$ |
| Spin $m_s$ | $\pm \tfrac12$ | — |
| Characteristic X-rays | $hf = (-13.6\text{ eV})(Z_\text{eff}^2/n_i^2 - Z_\text{eff}^2/n_f^2)$ | $Z_\text{eff} \approx Z-1$ for $K_\alpha$ |
Ch 29 Nuclear
| Quantity | Formula | Notes |
|---|---|---|
| Nucleus radius | $r = r_0 A^{1/3}$ | $r_0 = 1.2$ fm |
| Activity | $R = \lambda N$ | units: Bq = decays/s |
| Decay law | $N = N_0 e^{-\lambda t}$ | or $N = N_0(\tfrac12)^{t/T_{1/2}}$ |
| Half-life | $T_{1/2} = 0.693/\lambda$ | memorize the 0.693 |
| Q value | $Q = (m_\text{reactants} - m_\text{products})c^2$ | in MeV via $\times 931.494$ MeV/u |
| Threshold KE | $KE_\text{min} = (1 + m/M)|Q|$ | endothermic only |
Ch 30 Nuclear Energy
| Quantity | Formula | Notes |
|---|---|---|
| Energy released | $\Delta E = \Delta m \cdot c^2$ | $\Delta m$ = mass defect |
| Conservation | $\sum E_R(i) = \sum E_R(f) + \Delta E$ | all rest-energies |
| Conserve in reactions | $A$, $Z$, charge, energy, momentum | — |
Diffraction (Lab content, Ch 24 formulas)
| Quantity | Formula | Notes |
|---|---|---|
| Single-slit dark fringes | $y_m = m\lambda L/a$ | $m=1,2,3,\dots$ |
| Single-slit central width | $w_0 = 2\lambda L/a$ | twice the others |
| Single-slit higher widths | $w_m = \lambda L/a$ | — |
| Double-slit dark fringes | $y_m = (m+\tfrac12)\lambda L/d$ | $m=0,1,2,\dots$ |
| Double-slit fringe spacing | $\Delta y = \lambda L/d$ | uniform |
| $\lambda$ from single slit | $\lambda = w_0\cdot a/(2L)$ | solve for $\lambda$ |
| $\lambda$ from double slit | $\lambda = D_5 \cdot d/(9L)$ | $D_5$ = distance between $\pm 5^\text{th}$ darks |
10 Practice Problems — Click "Show solution" on each
Distribution mirrors professor's exam ratio: 3 Quantum · 3 Atomic · 2 Nuclear · 1 Nuclear Energy · 1 Diffraction.
Problem 1 Ch 27 · Photoelectric
Light of wavelength $\lambda = 250$ nm shines on a sodium surface (work function $\Phi = 2.30$ eV). (a) Find the maximum kinetic energy of the ejected photoelectrons in eV. (b) Find the stopping potential $V_s$. (c) What is the threshold wavelength $\lambda_0$ for sodium?
Show solution
(a) Photon energy via the shortcut:
$E_\text{photon} = hc/\lambda = 1240\ \text{eV}\cdot\text{nm} / 250\ \text{nm} = 4.96$ eV
$K_\text{max} = E_\text{photon} - \Phi = 4.96 - 2.30 = $ 2.66 eV
(b) $eV_s = K_\text{max} \Rightarrow V_s = K_\text{max}/e =$ 2.66 V
(c) $\lambda_0 = hc/\Phi = 1240/2.30 =$ 539 nm (yellow-green; longer wavelengths cannot eject electrons)
Problem 2 Ch 27 · Compton
An X-ray photon of wavelength $\lambda_0 = 0.0500$ nm scatters off a free electron at angle $\theta = 60°$. (a) What is the wavelength shift $\Delta\lambda$? (b) What is the scattered wavelength $\lambda'$? (c) What kinetic energy does the recoiling electron get (in keV)?
Show solution
(a) $\Delta\lambda = (h/m_e c)(1-\cos\theta) = (2.43\times10^{-12}\text{ m})(1 - \cos 60°)$
$= (2.43\times10^{-12})(0.5) = $ $1.215\times10^{-12}$ m = 0.001215 nm
(b) $\lambda' = \lambda_0 + \Delta\lambda = 0.0500 + 0.001215 = $ 0.05122 nm
(c) $K_e = E_0 - E' = hc/\lambda_0 - hc/\lambda'$
$= 1240/0.0500 - 1240/0.05122$ (in eV with $\lambda$ in nm)
$= 24800 - 24209 = 591$ eV ≈ 0.59 keV
Problem 3 Ch 27 · de Broglie
An electron is accelerated from rest through a potential difference of $V = 100$ volts. (a) What is its de Broglie wavelength in nm? (b) Compare to the size of a hydrogen atom ($\sim 0.1$ nm).
Show solution
(a) Use the shortcut $\lambda(\text{nm}) = 1.226/\sqrt{V}$:
$\lambda = 1.226/\sqrt{100} = 1.226/10 = $ 0.1226 nm
(Verify with the full formula: $p = \sqrt{2 m_e e V} = \sqrt{2\cdot 9.11\times10^{-31}\cdot 1.602\times10^{-19}\cdot 100} = 5.40\times10^{-24}$ kg·m/s; $\lambda = h/p = 6.63\times10^{-34}/5.40\times10^{-24} = 1.23\times10^{-10}$ m = 0.123 nm ✓)
(b) $\lambda \approx 0.12$ nm is comparable to atomic spacing (and to X-ray wavelengths). This is exactly the regime where electron diffraction works (Davisson–Germer 1927).
Problem 4 Ch 28 · Hydrogen spectrum
An electron in a hydrogen atom transitions from $n_i = 4$ to $n_f = 2$ (a Balmer-series line). (a) Find the energy of the emitted photon in eV. (b) Find the wavelength of the emitted photon in nm. What color is it?
Show solution
(a) $\Delta E = (-13.6\text{ eV})(1/n_i^2 - 1/n_f^2)$
$= (-13.6)(1/16 - 1/4)$
$= (-13.6)(0.0625 - 0.250)$
$= (-13.6)(-0.1875) = $ 2.55 eV (positive = photon emitted)
(b) $\lambda = 1240/\Delta E = 1240/2.55 = $ 486 nm
This is H$_\beta$, the cyan-blue Balmer line — clearly visible in hydrogen discharge tubes and stellar spectra.
Problem 5 Ch 28 · Bohr radius
Find the orbital radius of the electron in a hydrogen atom in the $n = 4$ excited state. Express in nm and compare to the ground-state radius.
Show solution
$r_n = (0.0529\text{ nm})\, n^2$
$r_4 = (0.0529)(4^2) = (0.0529)(16) = $ 0.846 nm
Ground state $r_1 = 0.0529$ nm. Ratio $r_4/r_1 = 16 = n^2$. The atom is 16× larger in physical size in the $n=4$ state.
Problem 6 Ch 28 · Characteristic X-ray
Find the wavelength of the $K_\alpha$ characteristic X-ray emitted by molybdenum ($Z = 42$). The transition is $n_i = 2 \to n_f = 1$, with $Z_\text{eff} = Z - 1$ for $K_\alpha$.
Show solution
$Z_\text{eff} = 42 - 1 = 41$.
$\Delta E = (-13.6\text{ eV})(Z_\text{eff}^2/n_i^2 - Z_\text{eff}^2/n_f^2) = (-13.6)(41^2)(1/4 - 1/1)$
$= (-13.6)(1681)(-0.75) = $ 17,150 eV ≈ 17.2 keV
$\lambda = 1240\ \text{eV}\cdot\text{nm}/17150\ \text{eV} = 0.0723$ nm = 0.0723 nm (≈ 72.3 pm)
(The accepted experimental value for Mo $K_\alpha$ is 0.0709 nm — within 2% of our $Z-1$ estimate.)
Problem 7 Ch 29 · Half-life
A sample contains $N_0 = 8.0\times10^{20}$ atoms of an isotope with half-life $T_{1/2} = 5.00$ years. How many atoms remain after 20.0 years?
Show solution
20.0 years = 4 half-lives.
$N = N_0 (\tfrac12)^{t/T_{1/2}} = (8.0\times10^{20})(\tfrac12)^4 = (8.0\times10^{20})(1/16) = $ $5.0\times10^{19}$ atoms
(Sanity check via the exponential: $\lambda = 0.693/5.00 = 0.1386$/yr; $N = 8.0\times10^{20}\cdot e^{-0.1386\cdot 20} = 8.0\times10^{20}\cdot e^{-2.772} = 8.0\times10^{20}\cdot 0.0625 = 5.0\times10^{19}$ ✓)
Problem 8 Ch 29 · Activity
A sample of $^{14}\text{C}$ contains $N = 1.00\times10^{12}$ atoms. Carbon-14 has a half-life $T_{1/2} = 5730$ years. (a) Find the decay constant $\lambda$ (per second). (b) Find the current activity $R$ in becquerels.
Show solution
(a) Convert half-life to seconds: $T_{1/2} = 5730 \cdot 365.25 \cdot 86400 \approx 1.808\times10^{11}$ s
$\lambda = 0.693/T_{1/2} = 0.693/(1.808\times10^{11}) = $ $3.83\times10^{-12}$ s$^{-1}$
(b) $R = \lambda N = (3.83\times10^{-12})(1.00\times10^{12}) = $ 3.83 Bq
(About 4 decays per second from a trillion atoms — that's why C-14 dating works on long timescales but needs precise counting equipment.)
Problem 9 Ch 30 · Mass-energy
The deuterium-tritium fusion reaction is $^{2}\text{H} + {}^{3}\text{H} \to {}^{4}\text{He} + n + \text{energy}$. Given the masses (in atomic mass units u):
- $m(^{2}\text{H}) = 2.01410$ u
- $m(^{3}\text{H}) = 3.01605$ u
- $m(^{4}\text{He}) = 4.00260$ u
- $m(n) = 1.00866$ u
How much energy (in MeV) is released per reaction?
Show solution
Mass of reactants: $2.01410 + 3.01605 = 5.03015$ u
Mass of products: $4.00260 + 1.00866 = 5.01126$ u
Mass defect: $\Delta m = 5.03015 - 5.01126 = 0.01889$ u
$\Delta E = \Delta m \cdot c^2 = (0.01889)(931.494\text{ MeV/u}) = $ 17.6 MeV
(Most of this — about 14.1 MeV — goes to the neutron's kinetic energy because it's much lighter than the He-4. This is what makes D-T fusion attractive for power but tricky for materials: those energetic neutrons are damaging to reactor walls.)
Problem 10 Lab · Diffraction
In the diffraction lab, you used a slit of width $a = 0.080$ mm at distance $L = 3.50$ m from the screen. You measured the central bright width $w_0 = 56.0$ mm. (a) Find the wavelength of the laser in nm. (b) Compute the percent error if the actual laser wavelength is 638 nm.
Show solution
(a) $w_0 = 2\lambda L/a$, so $\lambda = w_0 a/(2L)$.
$\lambda = (0.0560\text{ m})(0.080\times10^{-3}\text{ m}) / (2 \times 3.50\text{ m})$
$= (4.48\times10^{-6}) / 7.00 = 6.40\times10^{-7}$ m
$= $ 640 nm
(b) % error $= |640 - 638|/638 \times 100 = $ 0.31%
Great agreement — well within experimental uncertainty.
Cheat Sheet Setup Guide
You're allowed one hand-written 8.5×11 sheet. Below is a recommended layout. Print the template, then hand-copy the equations into the corresponding boxes on your real sheet. (The template's equations are pre-printed in light gray to give you a copy reference — you write over them in dark pen.)
⬇ Download Printable Template (PDF)Recommended layout — front side (4 quadrants)
- $E = hf = hc/\lambda$ (use $hc = 1240$ eV·nm)
- $K_\text{max} = hf - \Phi = eV_s$
- $\lambda_0 = hc/\Phi$
- $\lambda_\text{min} = hc/(e\Delta V)$
- $\lambda_\text{max} T = 2.898\times10^{-3}$ m·K
- $2d\sin\theta = m\lambda$
- $\Delta\lambda = (h/m_e c)(1-\cos\theta)$
- $\lambda = h/(mv)$ or $1.226/\sqrt{V}$ nm
- $\Delta x \Delta p \ge h/(4\pi)$
- $E_n = -13.6/n^2$ eV
- $r_n = 0.0529 n^2$ nm
- $hf = -13.6(1/n_i^2 - 1/n_f^2)$ eV
- Quantum numbers: $n, \ell, m_\ell, m_s$
- $\ell = 0,...,n-1$; $m_\ell = -\ell,...,+\ell$; $m_s = \pm\tfrac12$
- Pauli: no two e⁻ same set
- Char. X-ray: $hf = -13.6\, Z_\text{eff}^2(1/n_i^2 - 1/n_f^2)$
- $Z_\text{eff} \approx Z - 1$ for $K_\alpha$
- $r = r_0 A^{1/3},\ r_0 = 1.2$ fm
- $R = \lambda N$ (Bq)
- $N = N_0 e^{-\lambda t} = N_0(\tfrac12)^{t/T_{1/2}}$
- $T_{1/2} = 0.693/\lambda$
- $Q = (m_\text{in} - m_\text{out})c^2$
- $KE_\text{min} = (1 + m/M)|Q|$
- $1\text{ u} = 931.494$ MeV/$c^2$
- $\Delta E = \Delta m \cdot c^2$
- $\sum E_R(i) = \sum E_R(f) + \Delta E_\text{released}$
- Conserve $A$, $Z$, charge, energy, momentum
- DIFFRACTION:
- Single slit dark: $y_m = m\lambda L/a$
- Double slit dark: $y_m = (m+\tfrac12)\lambda L/d$
- $\lambda = w_0 a/(2L)$ or $D_5 d/(9L)$
Recommended layout — back side (overflow)
Use the back for constants & quick-recognition:
- Constants strip: $h = 6.63\times10^{-34}$ J·s, $c = 3.00\times10^{8}$ m/s, $hc = 1240$ eV·nm, $e = 1.60\times10^{-19}$ C, $m_e c^2 = 0.511$ MeV, $\lambda_C = 2.43$ pm, $r_0 = 1.2$ fm, $a_0 = 0.0529$ nm, 1 u = 931.494 MeV.
- Common work functions: Cs 1.9, Na 2.3, Cu 4.5, Pt 5.6 eV.
- Hydrogen series: Lyman ($n_f=1$, UV), Balmer ($n_f=2$, visible), Paschen ($n_f=3$, IR).
- Quantum numbers table: $n$ → any positive int; $\ell$ → 0...n−1; $m_\ell$ → −ℓ...+ℓ; $m_s$ → ±½.
- Quick-recognition cribsheet (see table below).
"When you see X, use formula Y"
| Trigger phrase | Formula to grab |
|---|---|
| "radiates", "stellar peak", "blackbody" | $\lambda_\text{max} T = 2.898\times10^{-3}$ |
| "photon energy", given $\lambda$ in nm | $E(\text{eV}) = 1240/\lambda(\text{nm})$ |
| "work function", "stopping potential" | $K_\text{max} = hf - \Phi = eV_s$ |
| "X-ray tube voltage", min $\lambda$ | $\lambda_\text{min} = hc/(e\Delta V)$ |
| "crystal", "diffraction angle" | $2d\sin\theta = m\lambda$ |
| "scattered off electron", angle $\theta$ | $\Delta\lambda = \lambda_C(1-\cos\theta)$ |
| "accelerated electron", de Broglie $\lambda$ | $\lambda(\text{nm}) = 1.226/\sqrt{V}$ |
| "hydrogen $n_i \to n_f$" | $\Delta E = 13.6(1/n_f^2 - 1/n_i^2)$ eV; $\lambda = 1240/\Delta E$ nm |
| "radius of hydrogen at $n$" | $r_n = 0.0529 n^2$ nm |
| "$K_\alpha$ X-ray", element $Z$ | Bohr with $Z_\text{eff} = Z - 1$ |
| "after $t$ years/half-lives" | $N = N_0(\tfrac12)^{t/T_{1/2}}$ |
| "activity in Bq", given $T_{1/2}$ and $N$ | $R = (0.693/T_{1/2})\cdot N$ |
| "energy released" in nuclear reaction | $\Delta E = \Delta m \cdot c^2 = \Delta m\cdot 931.494$ MeV |
| "single slit", "central bright" | $w_0 = 2\lambda L/a$ → $\lambda = w_0 a/(2L)$ |
| "double slit", $m^\text{th}$ dark | $y_m = (m+\tfrac12)\lambda L/d$ |
Cheat-sheet building tips
- Write small but legible. 0.5 mm gel pen, all-caps for headers helps.
- Group by chapter like the template — when you flip to "the Compton problem", you go straight to the Ch 27 quadrant.
- Color-code if your professor allows: one color for formulas, another for constants/units. Black-only is safest.
- Include units on at least the unusual constants ($k_B$, $\sigma$, $\lambda_C$ in pm, etc.).
- Rehearse finding things on the sheet during the practice problems above. If you can't find a formula in 5 seconds, the layout needs work.
Flashcards
Click the card to flip. Use ← → keys (or the buttons) to navigate.